A two-component therapy for adults with late-onset Pompe disease (LOPD) weighing 88 lbs or more who are not improving on their current enzyme replacement therapy (ERT).
POMBILITI + OPFOLDA resetting expectations
JARED
DIAGNOSED WITH LOPD IN 1998
POMBILITI + OPFOLDA WAS SHOWN TO IMPROVE WALKING DISTANCE AND BREATHING FUNCTION VS THE COMPARATOR* IN ERT-EXPERIENCED ADULTS
Study Design
POMBILITI + OPFOLDA was evaluated in a randomized, controlled clinical study vs the Comparator* over 52 weeks.
123 PEOPLE TOTAL IN THE STUDY, INCLUDING:
ERT-experienced
People in this group were on an ERT for at least 2 years
ERT-naïve †
People in this group had never received an ERT
85 people received POMBILITI + OPFOLDA
38 people received the Comparator
- The study’s primary endpoint (goal) was to see how much farther people who received POMBILITI + OPFOLDA could walk in 6 minutes, using the 6-Minute Walk Test (6MWT)‡
- Forced vital capacity (FVC), a test of breathing ability, was a key secondary endpoint (goal) of the study
- ERT-experienced people were on treatment for an average of 7.4 years before the study
*An alglucosidase alfa product not approved in the US + placebo.
†POMBILITI + OPFOLDA is not approved for use in ERT-naïve people.
‡Results of the 6MWT were numerically favorable but did not meet the primary endpoint of statistical superiority for the full study population.
DISCOVER THE DATA
Watch this video for a breakdown of the clinical trial and safety results
Improvements in
Walking Distance
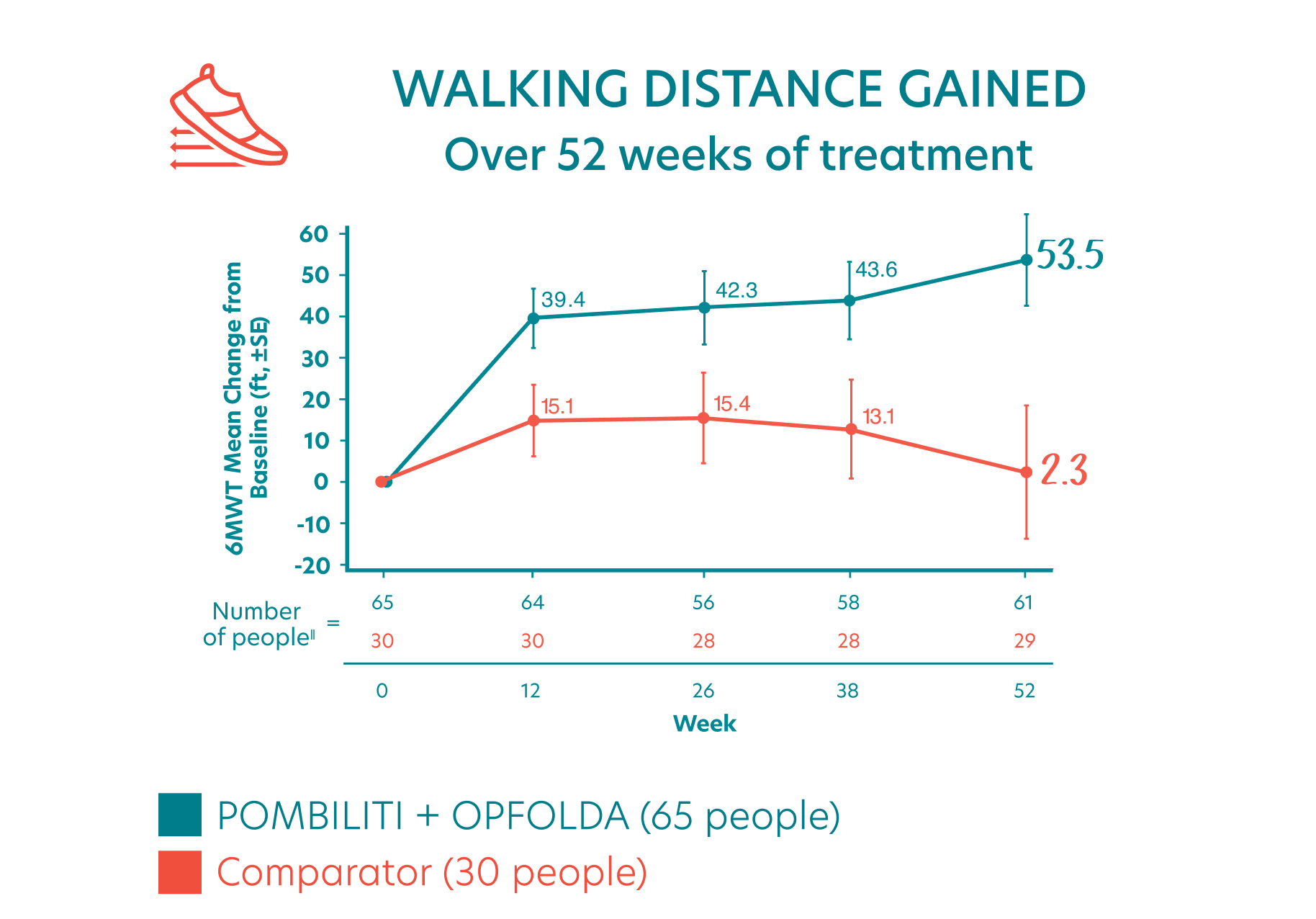
Study participants were assessed using the 6MWT, a timed test to measure how far a person can walk in 6 minutes.
ERT-experienced adults receiving POMBILITI + OPFOLDA had IMPROVED WALKING DISTANCE versus adults receiving the Comparator§
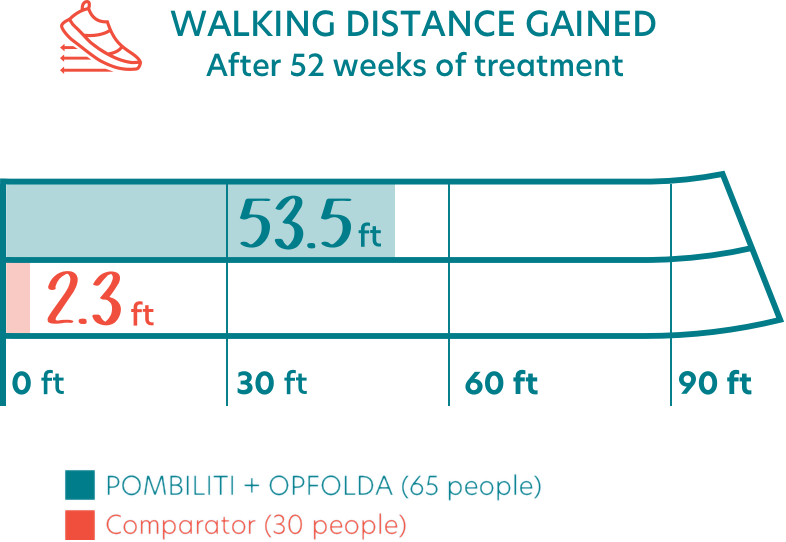
- People who received POMBILITI + OPFOLDA walked an average of 53.5 feet farther from the start of the study
- People who received the Comparator walked an average of 2.3 feet farther from the start of the study
- This indicated an estimated treatment effect of more than 50 feet for POMBILITI + OPFOLDA versus the Comparator
§Results of the 6MWT and FVC for ERT-experienced people were numerically favorable but were not tested to determine statistical superiority of POMBILITI + OPFOLDA vs the Comparator.
‖The number of people for each time point in the chart changed because some people missed some of the tests and some people did not complete the study.
Improvements in
Breathing
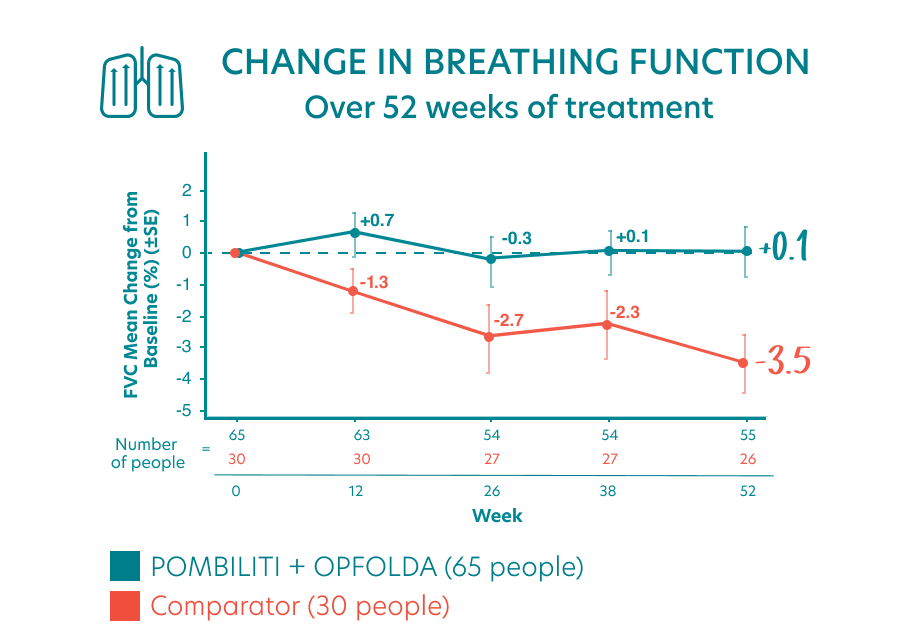
Study participants were assessed using FVC, a test of breathing ability.
ERT-experienced adults receiving POMBILITI + OPFOLDA had IMPROVED BREATHING FUNCTION versus adults receiving the Comparator§
- People who received POMBILITI + OPFOLDA who had previous experience with ERT saw their breathing increase by an average of 0.1% from the start of the study
- ERT-experienced adults who received the Comparator saw their breathing decline by an average of 3.5% from the start of the study
- This indicated an estimated treatment effect of 3.5% for POMBILITI + OPFOLDA
§Results of the 6MWT and FVC for ERT-experienced people were numerically favorable but were not tested to determine statistical superiority of POMBILITI + OPFOLDA vs the Comparator.
||The number of people for each time point in the chart changed because some people missed some of the tests and some people did not complete the study.
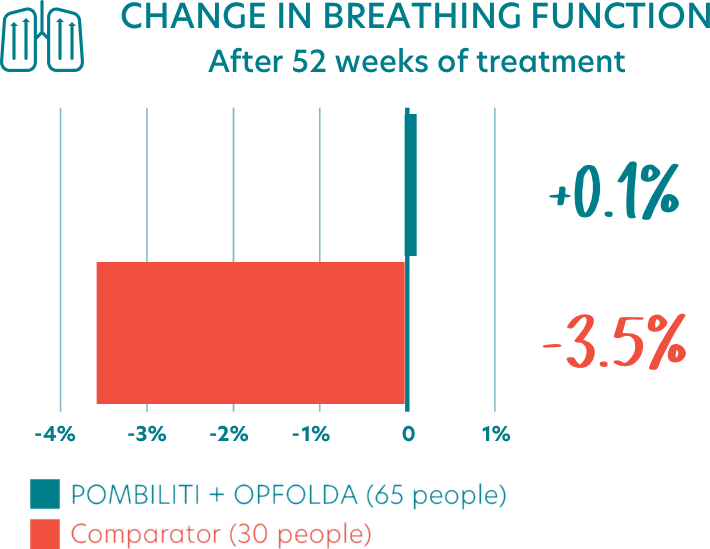
Biomarker Data: Improvements in
Measures of Glycogen Buildup
Study participants were tested for Hex4, a measure of glycogen buildup.
ERT-experienced adults receiving POMBILITI + OPFOLDA had IMPROVED MEASURES OF GLYCOGEN BUILDUP
- Lower Hex4 test results are a sign that there is less glycogen accumulation
- People receiving POMBILITI + OPFOLDA saw an average reduction of 29% in Hex4 vs an average increase of 20% with the Comparator
Safety
Information about side effects with POMBILITI + OPFOLDA
The most common side effects (≥5%) reported during the phase 3 PROPEL study were headache and diarrhea.
The most common side effects reported in at least 5% of participants treated with POMBILITI + OPFOLDA across 3 clinical trials were headache, diarrhea, fatigue, nausea, abdominal pain, and fever.
Serious adverse reactions reported in 2 or more people treated with POMBILITI + OPFOLDA were severe allergic reaction and hives. A total of 5 people treated with POMBILITI + OPFOLDA permanently discontinued POMBILITI due to side effects, including 4 who discontinued the treatment because of a serious side effect.
Additional side effects
Side effects reported in at least 2% of adults treated with POMBILITI + OPFOLDA across 3 clinical trials included: muscle aches, joint pain, increased blood pressure, pain, tremor (shaking), indigestion, weakness, constipation, infusion site swelling, flank (side) pain, feeling unwell, burning or pins and needles feeling, drowsiness, and decreased platelet count.
POMBILITI in combination with OPFOLDA may cause serious side effects, including: hypersensitivity reactions (including anaphylaxis), infusion-associated reactions, and risk of acute cardiorespiratory failure. Please see IMPORTANT SAFETY INFORMATION.
These are not all of the possible side effects of POMBILITI in combination with OPFOLDA.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.

“By the end of the 1-year clinical study, my performance on 6MWT and FVC has stabilized. These results are important to me.”
—
Jared on making the switch to POMBILITI + OPFOLDA

