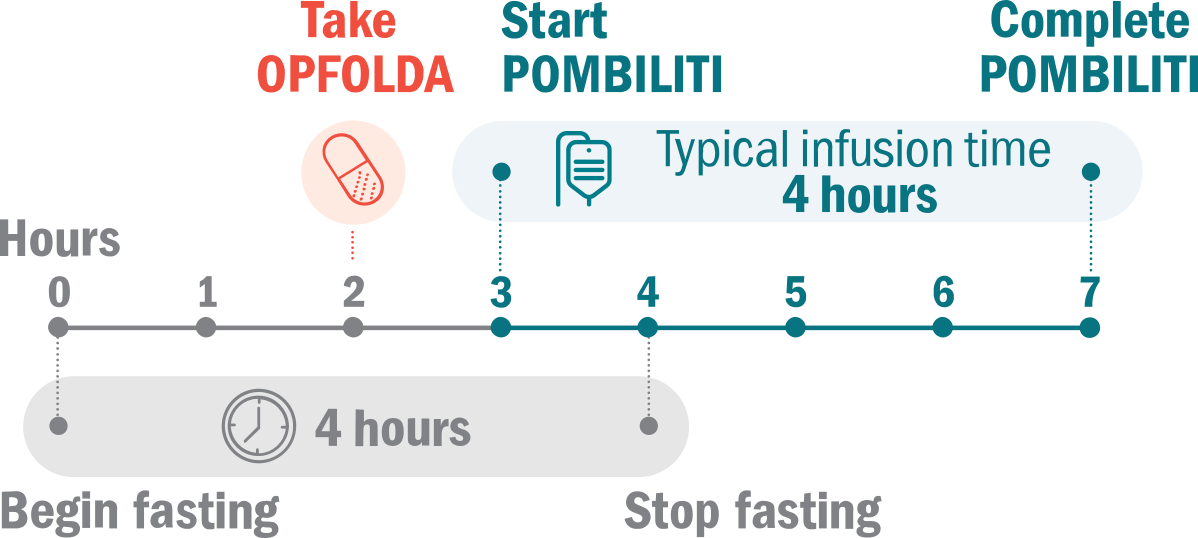
A two-component therapy for adults with late-onset Pompe disease (LOPD) weighing 88 lbs or more who are not improving on their current enzyme replacement therapy (ERT).

Support+Resourceshelping you manage LOPD
SEE HOW IT ALL COMES TOGETHER
To understand how POMBILITI + OPFOLDA work together, watch this video
To explore the clinical trial results for POMBILITI + OPFOLDA, watch this video
Downloadable Resources

PRODUCT BROCHURE

TREATMENT DAY GUIDE

INSURANCE 101 BROCHURE
Get started
Discuss
Talk to your doctor to see if POMBILITI + OPFOLDA could be right for you.
Download the Discussion Driver to help prepare for upcoming conversations with your care team.
PREPARE
Learn what to expect and get help managing your treatment day experience.
Download the MyDay Pompe™ app from the App Store or Google Play.
Apple, the Apple logo, and iPhone are trademarks of Apple Inc., registered in the U.S. and other countries and regions. App Store is a service mark of Apple Inc. Google Play and the Google Play logo are trademarks of Google LLC.
Connect
Find support.
AMICUS ASSIST® provides comprehensive support to people prescribed POMBILITI + OPFOLDA. A Patient Education Liaison (PEL) and a Case Manager are standing by to help.
Get in touch with us by calling 1-833-AMICUS-A (1-833-264-2872).

Enjoy our AMICUS ASSIST Spotify® playlists.
Music. Meditations. Audiobooks. We've put together selections just for you. They can help you stay entertained, relax, or even learn something new.
Click the buttons to stream these free Spotify playlists.
The Spotify logo is a trademark of Spotify LLC.
The following Pompe disease organizations and websites can help you connect with others while providing information that may be relevant to your experience.

Acid Maltase Deficiency Association (AMDA)
The AMDA assists in funding research and promoting public awareness of Pompe disease.
Visit site
International Pompe Association
The IPA is a federation of Pompe disease patient groups worldwide that seeks to coordinate activities and share experience and knowledge among different international organizations.
Visit site
Pompe Alliance
Pompe Alliance provides supportive services, education, and information to patients, caregivers, medical professionals, and Pompe community stakeholders.
Visit site
Pompe Warrior Foundation (PWF)
The PWF promotes research, educates, and empowers individuals and families affected by Pompe disease and other lysosomal storage diseases.
Visit site
United Pompe Foundation (UPF)
The UPF assists patients and/or their families with medical costs and other expenses they face and may not be able to cover, or fully cover, through their insurance.
Visit siteStill have questions? We’ve got answers in our Frequently Asked Questions.
Get connected
Sign up to receive more information about LOPD and Pombiliti + Opfolda.
Important Safety Information
POMBILITI in combination with OPFOLDA may cause serious side effects, including:
- Hypersensitivity reactions (including anaphylaxis): Severe and potentially life-threatening allergic-type reactions related to the infusion may occur during and after POMBILITI in combination with OPFOLDA treatment. Your doctor will inform you of the signs and symptoms of hypersensitivity reactions which may include: difficulty breathing or swallowing, rash or hives, low blood pressure, swelling of lips, tongue, throat or face. Seek medical care immediately should signs and symptoms occur. If a severe reaction occurs, your doctor may decide to immediately discontinue the infusion and provide medical care. Appropriate medical support measures may be administered, and you may require close observation during and after POMBILITI infusion.
What are POMBILITI and OPFOLDA?
POMBILITI and OPFOLDA are prescription medicines used in combination for the treatment of adults with late-onset Pompe disease weighing 88 pounds (40 kg) or more and who are not improving on their current enzyme replacement therapy (ERT).
It is not known if POMBILITI in combination with OPFOLDA is safe and effective in children with late-onset Pompe disease.
Important safety information
POMBILITI in combination with OPFOLDA may cause serious side effects, including:
- Hypersensitivity reactions (including anaphylaxis): Severe and potentially life-threatening allergic-type reactions related to the infusion have been reported during and after POMBILITI in combination with OPFOLDA treatment. Your doctor will inform you of the signs and symptoms of hypersensitivity reactions which may include: difficulty breathing or swallowing; rash or hives; low blood pressure; swelling of lips, tongue, throat, or face. Seek medical care immediately should signs and symptoms occur. If a severe reaction occurs, your doctor may decide to immediately discontinue the infusion and provide medical care. Appropriate medical support measures may be administered, and you may require close observation during and after POMBILITI infusion.
- Infusion-Associated Reactions (IARs): Severe IARs related to the infusion have been reported during or after POMBILITI in combination with OPFOLDA. Your doctor will inform you of the signs and symptoms of hypersensitivity reactions which may include: hives, itching, shortness of breath, flushing, chills, and low blood pressure. Seek medical care immediately should signs and symptoms occur. If severe IARs occur during infusion, your doctor may decide to immediately discontinue the infusion and provide appropriate medical care. If you have an acute underlying illness at the time of POMBILITI infusion you may be at greater risk for IARs. If you have advanced Pompe disease you may have compromised heart and breathing function, which may put you at a higher risk of severe complications from IARs.
- Risk of Acute Cardiorespiratory Failure: If you are likely to develop fluid volume overload or have an acute breathing condition or heart and/or breathing problems that require fluid restriction, you may be at risk of worsening of your heart or breathing status during POMBILITI infusion. Your doctor may decide that close observation during and after POMBILITI administration may be necessary.
Do not take POMBILITI in combination with OPFOLDA if you are pregnant.
Before taking POMBILITI in combination with OPFOLDA, tell your healthcare provider about all of your medical conditions, including if you:
- have kidney problems
- are pregnant or plan to become pregnant. POMBILITI in combination with OPFOLDA may cause harm to your unborn baby.Females who are able to become pregnant:
- Your healthcare provider will check if you are pregnant before you start treatment with POMBILITI in combination with OPFOLDA.
- You should use effective birth control (contraception) during treatment with POMBILITI in combination with OPFOLDA and for at least 60 days after the last dose.
- Tell your healthcare provider right away if you become pregnant or think you might be pregnant during treatment with POMBILITI in combination with OPFOLDA.
- are breastfeeding or plan to breastfeed. It is not known if OPFOLDA alone or in combination with POMBILITI passes into your breast milk. Do not breastfeed during treatment with POMBILITI in combination with OPFOLDA. Talk to your healthcare provider about the best way to feed your baby during this time.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
POMBILITI and OPFOLDA must be taken in combination. POMBILITI in combination with OPFOLDA will be given to you 1 time every other week.
The most common side effects of POMBILITI in combination with OPFOLDA include: headache, diarrhea, tiredness, nausea, stomach area pain, and fever.
POMBILITI in combination with OPFOLDA may cause fertility problems in females and males, which may affect the ability to have children. Talk to your healthcare provider if you have concerns about fertility.
These are not all the possible side effects of POMBILITI and OPFOLDA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
What are POMBILITI and OPFOLDA?
POMBILITI and OPFOLDA are prescription medicines used in combination for the treatment of adults with late-onset Pompe disease weighing 88 pounds (40 kg) or more and who are not improving on their current enzyme replacement therapy (ERT).
It is not known if POMBILITI in combination with OPFOLDA is safe and effective in children with late-onset Pompe disease.
Please see full Prescribing Information, including BOXED WARNING, for POMBILITI and full Prescribing Information and Patient Information for OPFOLDA.